Ice Phenomena That Defy Expectations
Ice seems simple. Water gets cold, turns solid, and you get ice. But the reality is far stranger than that basic description suggests.
Ice does things that contradict common sense, breaks the rules you learned in school, and creates natural wonders that seem impossible until you see them yourself. Some of these phenomena happen in your freezer.
Others require specific conditions found only in remote corners of the planet. All of them challenge what you think you know about frozen water.
Ice Expands When It Freezes

Most materials shrink when they freeze. Molecules pack together more tightly in a solid state than in a liquid.
That’s how physics typically works. Ice does the opposite. When water freezes, it expands by about 9%.
The molecular structure of ice takes up more space than liquid water. This is why ice floats—it’s less dense than the water around it.
This single property changed everything. If ice sank like most solids, lakes and oceans would freeze from the bottom up.
Aquatic life as we know it couldn’t exist. The entire ecosystem of Earth would be completely different.
One weird quirk of water’s molecular structure made complex life possible.
Hot Water Freezes Faster Than Cold Water

This sounds like it violates basic thermodynamics. Hot water has more heat to lose before freezing, so it should take longer.
Logic says cold water wins the race to freeze. Except sometimes hot water actually does freeze faster.
Scientists call this the Mpemba effect, named after a Tanzanian student who noticed it while making ice cream in the 1960s. The exact mechanism still puzzles researchers, but it happens reliably under certain conditions.
Several factors contribute. Hot water evaporates faster, reducing the volume that needs to freeze.
Hot water can create convection currents that distribute cold more efficiently. The containers might matter.
The dissolved gases might matter. Nobody has a complete explanation, but the phenomenon is real.
You can test this yourself. Put two identical containers of water in your freezer—one hot, one cold.
Sometimes the hot one freezes first. Not always, but often enough to be weird.
Ice Can Form Without Water

Dry ice isn’t really ice at all. It’s frozen carbon dioxide that skips the liquid phase entirely, going straight from gas to solid. At normal atmospheric pressure, carbon dioxide can’t exist as a liquid—it sublimates directly into gas.
But carbon dioxide isn’t the only substance that creates ice-like solids. Methane, ammonia, and nitrogen all form ices under the right conditions.
These exotic ices exist on distant moons and planets where temperatures drop to extremes Earth never reaches. Titan, Saturn’s largest moon, has lakes and seas made of liquid methane and ethane.
When these liquids freeze, they create methane ice—a substance that behaves differently from water ice in almost every way. It’s denser than the liquid it forms from, so it sinks.
The entire geology of alien worlds operates on rules that seem wrong based on what we know from Earth.
Ice Flows Like a Glacier Flows Like a River

Glaciers move. Everyone knows that.
But ice is solid—how does solid ice flow? The answer defies intuition. Under enough pressure, ice behaves as a plastic material.
The immense weight of a glacier causes the ice at the bottom to deform and flow, even though it stays frozen solid. Ice crystals slip past each other at a molecular level, allowing the entire mass to move downhill like an extremely slow river.
The top of a glacier stays rigid and brittle. The bottom flows.
This creates strange effects. Crevasses form on the surface while the base slides along bedrock.
Objects dropped on a glacier don’t stay in one spot—they travel with the flowing ice, sometimes emerging decades later miles away from where they entered.
Supercooled Water Stays Liquid Below Freezing

Water doesn’t have to freeze at 0°C. Under the right conditions, you can cool it well below freezing and it stays liquid.
Scientists call this supercooled water, and it’s far more common than you’d think. Clouds often contain supercooled water droplets at temperatures of -10°C or colder.
These droplets stay liquid because they lack nucleation sites—tiny imperfections or particles that give ice crystals a place to start forming. The water wants to freeze, but it needs something to trigger the phase change.
Touch supercooled water and it freezes instantly. Disturb it and ice crystals spread through the liquid in seconds.
The transformation happens so fast that you can watch it happen. Bottles of purified water sometimes supercool in your freezer, staying liquid until you open them or tap the bottle.
Aircraft flying through clouds with supercooled water face serious danger. The droplets freeze instantly on contact with the plane’s surface, forming ice that disrupts airflow and adds weight.
This icing can bring down aircraft if pilots don’t recognize it quickly enough.
Ice Can Burn You
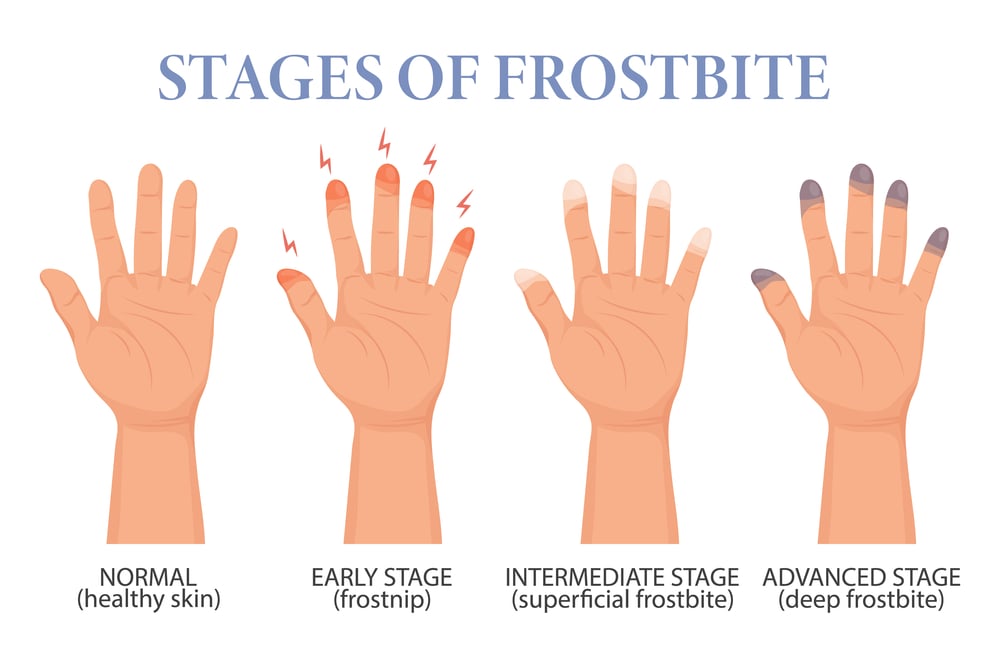
Frostbite happens when tissue freezes, but that’s not what makes ice dangerous to touch. The real danger is how fast ice pulls heat from your skin.
Ice at 0°C won’t hurt you much if you touch it briefly. But ice colder than that—especially dry ice at -78°C—causes instant damage.
The extreme cold doesn’t just freeze your skin. It causes cold burns that damage tissue the same way heat burns do.
Your body can’t sense the difference between extreme cold and extreme heat. Both trigger the same pain receptors.
This is why dry ice feels like it’s burning when you touch it. Your nerves detect tissue damage and interpret it as burning, even though the mechanism is completely opposite to thermal burns.
Ice Makes Bizarre Geometric Patterns

When water freezes slowly enough, it creates perfect geometric structures. Snowflakes form six-sided crystals because of how water molecules bond together.
The hexagonal symmetry is built into ice’s molecular structure. But ice creates other geometric patterns too. Ice crystals can form needles, plates, columns, and dendrites depending on temperature and humidity.
The shapes follow precise mathematical rules. Change the temperature by a few degrees and the crystal structure completely changes.
Frost on your window follows similar rules. The patterns aren’t random—they’re determined by microscopic imperfections in the glass and the exact conditions when the water vapor froze.
Each frost pattern is unique, but they all follow the same geometric principles.
Ice Cores Store Ancient Air

Glaciers trap tiny bubbles of air as snow compresses into ice. These bubbles preserve samples of the atmosphere from when the ice formed.
Some ice cores contain air that’s hundreds of thousands of years old. Scientists drill into Antarctic ice and extract cores that tell them what Earth’s atmosphere was like before humans existed.
The bubbles contain the exact mixture of gases present at the time, including carbon dioxide, methane, and oxygen levels. It’s like having a time capsule that preserves not just objects but the actual air from the past.
The oldest ice cores go back 800,000 years. They show how Earth’s climate changed naturally over time, revealing patterns of ice ages and warm periods.
This data helps scientists understand current climate change by providing context for what’s normal variation and what’s unprecedented.
Black Ice Forms Where You Least Expect It

Black ice isn’t actually black. It’s perfectly clear ice that forms on roads, making it nearly invisible.
The dark pavement shows through, which is why people call it black ice. This ice forms when temperatures hover right around freezing and moisture on the road freezes in a thin, smooth layer. Rain, fog, or even humidity can create it.
The ice is so thin and clear that drivers don’t see it until they’re already sliding. Black ice defies expectations because roads can have it even when the air temperature is above freezing.
Bridges and overpasses freeze first because cold air circulates underneath them. Shaded areas can have ice while sunny spots are clear.
You can’t trust your eyes—the road that looks wet might actually be ice.
Ice Can Explode

Freezing water expands with tremendous force. When water freezes inside a confined space, the pressure builds until something breaks.
Pipes burst in winter because ice forms inside them and the expansion splits the metal or plastic. But ice can do more dramatic things than burst pipes. Rocks can shatter when water seeps into cracks and freezes.
The expansion creates pressure that exceeds the rock’s strength. This process, called frost wedging, breaks apart mountains over time.
In rare cases, ice can actually explode. When methane or other gases get trapped in ice under pressure, disturbing the ice can release the gas suddenly.
The ice shatters and the gas escapes with enough force to create an explosion. This happens sometimes in frozen lakes where methane bubbles get trapped in ice.
Ice Has at Least 18 Different Structures

The ice in your freezer is called Ice Ih. The “I” means it’s the first type discovered, and the “h” indicates its hexagonal crystal structure.
But Ice Ih is just one form of ice—scientists have identified at least 18 different crystalline structures of ice. These exotic forms of ice only exist under extreme conditions of pressure and temperature. Ice II forms under high pressure and has a different crystal structure than normal ice. Ice III, Ice IV, Ice V, and so on each have their own unique properties.
Some are denser than water, unlike Ice Ih. These exotic ices probably exist inside planets and moons.
The interiors of icy bodies like Europa and Enceladus contain conditions that could create high-pressure forms of ice. Understanding these structures helps scientists model what’s inside distant worlds, even though we’ll never see these exotic ices on Earth’s surface.
Ice Spikes Form in Your Freezer

Sometimes when you freeze water in an ice cube tray, a spike of ice grows upward from the surface. These ice spikes can be several inches tall, like little frozen mountains rising from the cube.
This happens because ice forms from the outside in. The surface freezes first, creating a shell.
As the water inside continues freezing and expanding, the pressure pushes unfrozen water up through a small pit in the ice shell. This water freezes as it emerges, creating a spike.
The process requires specific conditions—the water needs to be pure enough, the freezing needs to happen at the right rate, and there needs to be a small opening in the ice shell. Most of the time these conditions don’t align. But when they do, you get these weird spiky ice structures that look like they shouldn’t exist.
Hair Ice Grows on Dead Wood

In certain forests during winter, dead wood sprouts what looks like fine white hair. These delicate ice filaments can be several centimeters long and as fine as human hair.
They appear overnight and melt by morning. Scientists puzzled over hair ice for more than a century.
The ice grows perpendicular to the wood surface in thin strands that look nothing like normal frost. The mystery deepened because hair ice only forms on certain types of dead wood.
Researchers finally solved it in 2015. A fungus living in the wood creates the conditions needed for hair ice to form.
The fungus modifies how water freezes, allowing it to form these delicate strands instead of normal ice crystals. Without the fungus, the wood just gets regular frost.
This means hair ice is technically a collaboration between ice and a living organism. The phenomenon requires both to exist, making it one of the strangest interactions between life and frozen water.
When Water Refuses to Obey

Strange how ice behaves. When cold hits, it swells instead of shrinking like everything else would.
Floating seems wrong for a solid – yet there it is, sitting on water. Some versions hide in labs, shaped by pressures not found outside experiments.
The kind you make at home? Breaks logic without trying. Not just odd details – these mismatches shape the planet’s core mechanics.
Because ice rises, living things survive. Storm rhythms form when cloud frost builds a certain way.
Old skies stay locked inside glaciers, letting us trace past climates. Starting with a frosty fingertip – there’s something odd going on.
Ice feels common, sure, but only due to constant run-ins. Actually, this stuff breaks expectations every single time.
Physics expects one thing, yet here comes ice doing the opposite again.
More from Go2Tutors!

- The Romanov Crown Jewels and Their Tragic Fate
- 13 Historical Mysteries That Science Still Can’t Solve
- Famous Hoaxes That Fooled the World for Years
- 15 Child Stars with Tragic Adult Lives
- 16 Famous Jewelry Pieces in History
Like Go2Tutors’s content? Follow us on MSN.



