Unusual Chemicals You’ve Never Heard Of
The world of chemistry holds countless surprises. Most people know about common substances like salt and sugar, but the laboratory shelves are packed with compounds that sound like they belong in a fantasy novel. These chemicals often have bizarre properties, strange names, or fascinating backstories that make them far more interesting than their everyday cousins.
Ready to discover some of chemistry’s best kept secrets? These compounds will change how you think about the molecular world around us.
Buckminsterfullerene

Named after the famous architect Buckminster Fuller, this carbon compound looks exactly like a tiny soccer game piece. It has a cage-like fused-ring structure (truncated icosahedron) made of twenty hexagons and twelve pentagons, and resembles a football.
The discovery of this molecule revolutionized chemistry because nobody expected carbon atoms to arrange themselves in such a perfect hollow sphere. Scientists can actually trap other atoms inside these carbon cages, creating materials with entirely new properties.
Ferrocene

This bright orange compound smells surprisingly like mothballs. The molecule is a complex consisting of two cyclopentadienyl rings sandwiching a central iron atom.
What makes ferrocene truly special is its sandwich structure, where an iron atom sits perfectly centered between two ring-shaped carbon structures. This discovery helped scientists understand how metals could bond with organic molecules in ways nobody thought possible before.
Like Go2Tutors’s content? Follow us on MSN.
Astatine
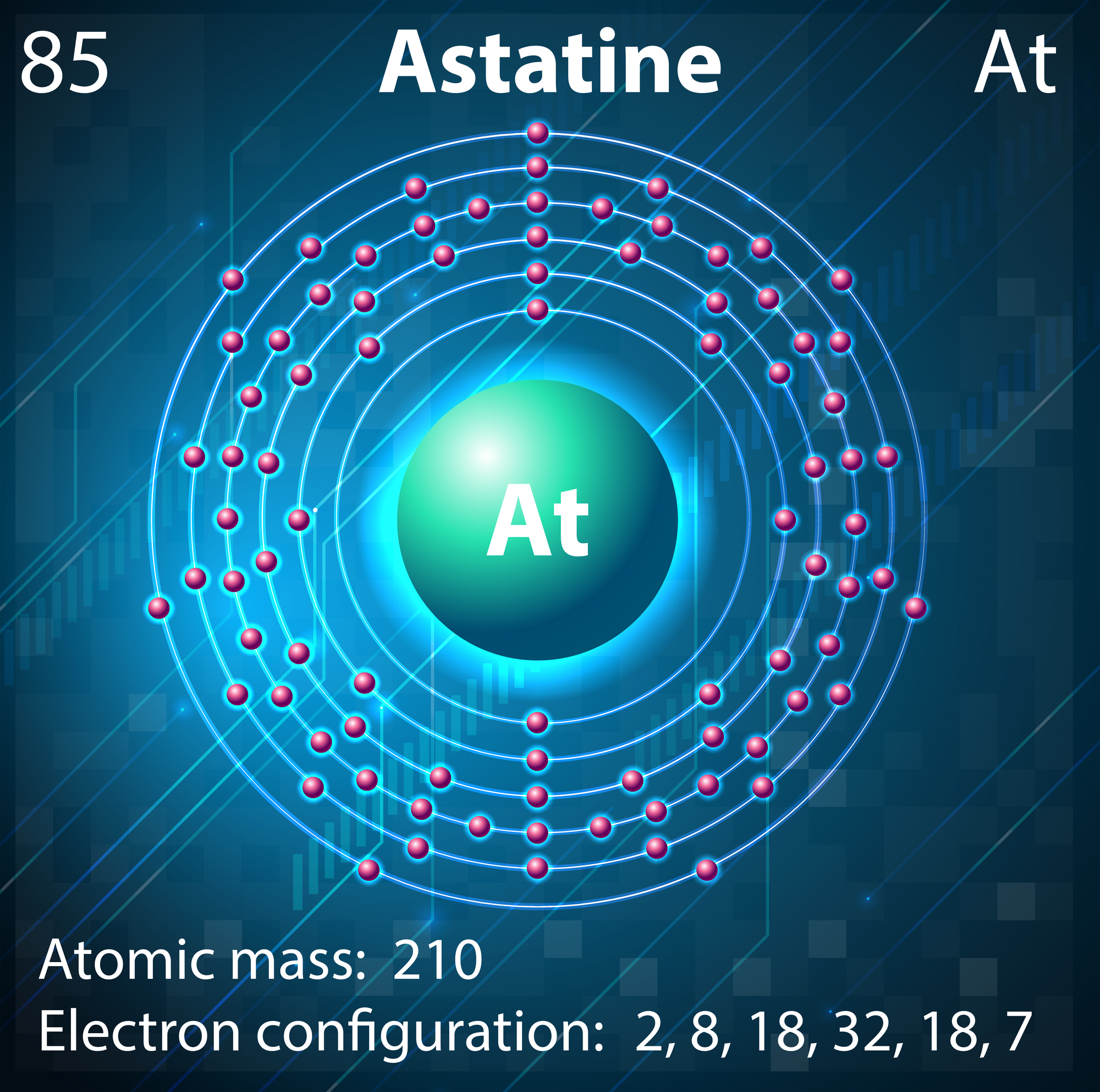
A solid sample of the element has never been seen, because any macroscopic specimen would be immediately vaporized by the heat of its radioactivity. This element is so unstable that it destroys itself faster than scientists can study it properly.
Even though astatine appears on the periodic table, getting enough to actually see requires incredible luck and timing. The few atoms that scientists have managed to create disappear within hours.
Adamantane
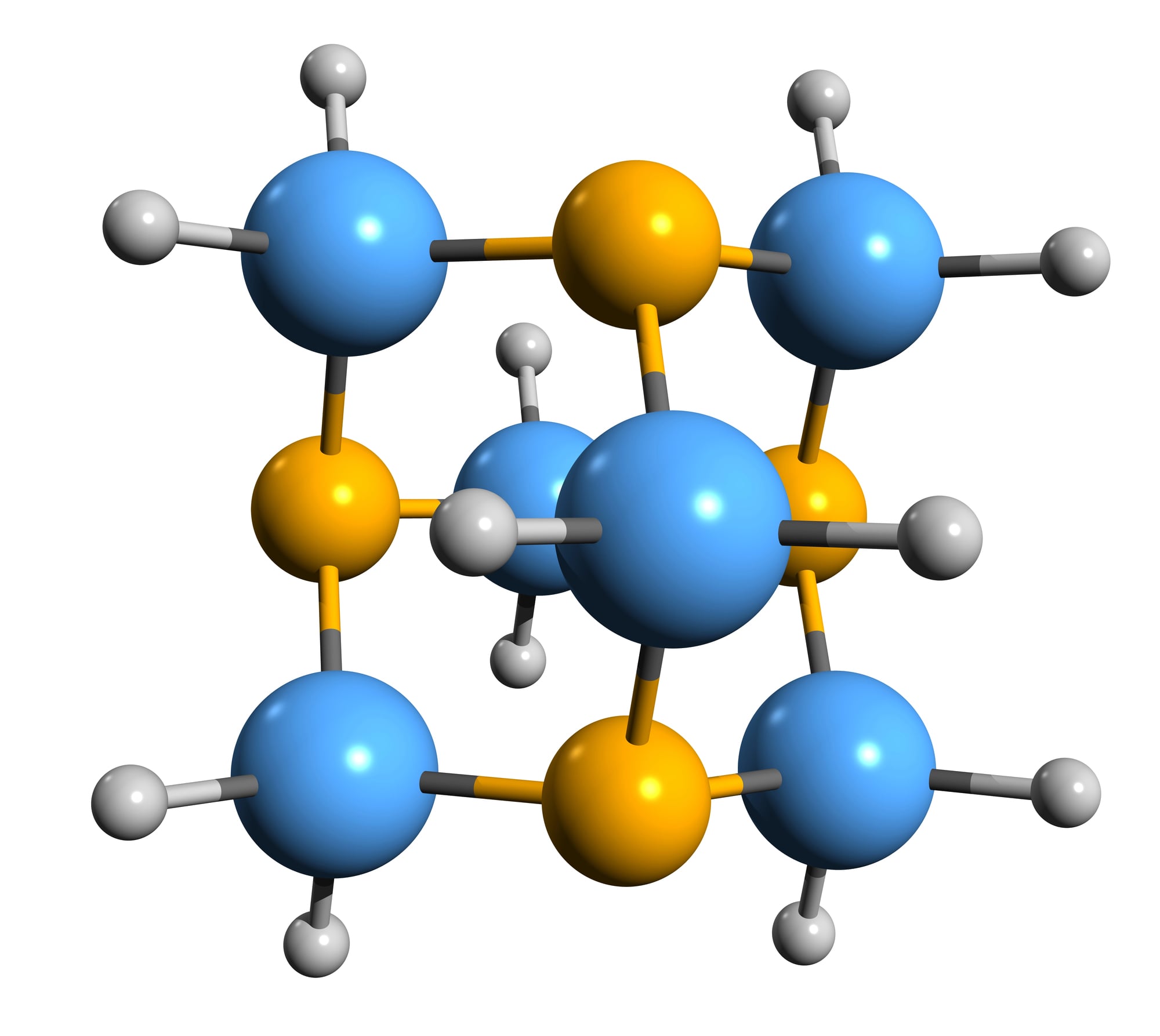
The name comes from diamond, and for good reason. This carbon compound has the same structure as tiny pieces of diamond, making it incredibly stable and rigid.
The research into adamantane-type compounds has gained momentum in recent years, yielding remarkable new applications for this class of materials. Scientists use adamantane as a building block for creating new materials that need to be both lightweight and incredibly strong.
Methyl anthranilate
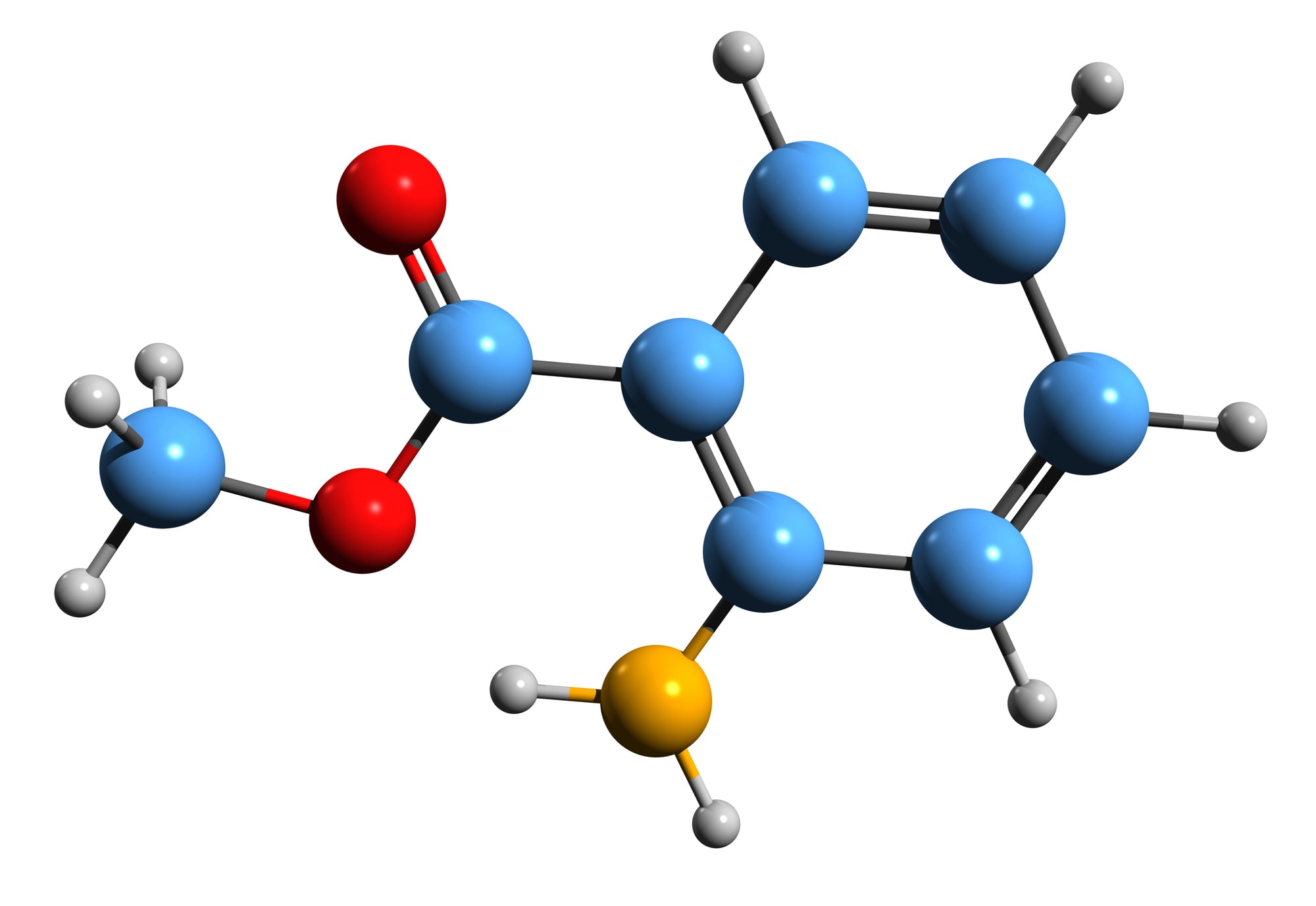
Turns out it smelled like grapes because it’s the actual reason grapes smell like grapes. This chemical gives grapes their distinctive smell and flavor, yet most people think artificial grape flavor tastes nothing like real grapes.
The irony is that this compound is what makes grapes taste like grapes in the first place. Food scientists discovered it by accident while trying to create better artificial flavors.
Like Go2Tutors’s content? Follow us on MSN.
Thulium

This rare earth element is so uncommon that most chemistry textbooks barely mention it. These elements tell a deeper story, one of continuous discovery, relentless curiosity, and the ever-expanding boundaries of human knowledge.
Thulium costs more than gold and has very few practical uses, but its rarity makes it fascinating to scientists who study the limits of what nature can create.
Francium

The rarest naturally occurring element on Earth has some mind-bending properties. Only about one ounce of francium exists on the entire planet at any given time.
This element is so radioactive that it glows from its own energy, but finding enough to actually see that glow is nearly impossible. Scientists estimate that all the francium atoms in your body would fit on the head of a pin.
Californium

Named after the state where it was first created, californium is one of the most expensive materials on Earth. A single gram costs about 25 million dollars to produce.
This artificial element has neutron-emitting properties that make it useful for detecting explosives and oil deposits. Only a few laboratories in the world can make it, and the entire global supply would fit in a small jar.
Like Go2Tutors’s content? Follow us on MSN.
Aerogel

This substance looks like solid smoke and feels like touching a cloud. Aerogel is mostly empty space, making it lighter than air itself while still maintaining a solid structure.
Scientists use it to capture particles from space because nothing else can trap such tiny, fast-moving objects without destroying them. Despite being incredibly light, aerogel can support thousands of times its own weight.
Gallium

This metal melts in your hand like ice cream on a hot day, but it has a darker side. Gallium can destroy aluminum by making it incredibly brittle, which is why it’s banned from many aircraft.
The metal stays liquid at room temperature and can be molded like clay, yet it becomes hard as steel when cooled. Scientists use gallium in electronics because of its unique properties.
Looking back to understand today’s world

These unusual chemicals remind us that the natural world operates by rules far stranger than most people imagine. What started as curious laboratory discoveries often becomes tomorrow’s breakthrough technology.
The same compounds that once seemed like chemistry’s odd cousins now power our electronics, protect our spacecraft, and save lives in hospitals. Understanding these unusual substances helps us appreciate just how much mystery still lies hidden in the molecular world around us.
Like Go2Tutors’s content? Follow us on MSN.
More from Go2Tutors!

- 16 Historical Figures Who Were Nothing Like You Think
- 12 Things Sold in the 80s That Are Now Illegal
- 15 VHS Tapes That Could Be Worth Thousands
- 17 Historical “What Ifs” That Would Have Changed Everything
- 18 TV Shows That Vanished Without a Finale
Like Go2Tutors’s content? Follow us on MSN.



