Most Toxic Chemical Compounds Created in Modern Labs
The pursuit of scientific knowledge has led researchers down some remarkably dangerous paths. Modern chemistry labs have synthesized compounds so lethal that a single misstep could prove catastrophic.
These substances exist at the extreme edge of chemical toxicity, created not for destruction, but for understanding the fundamental limits of molecular behavior. Each represents years of careful research conducted under the most stringent safety protocols imaginable.
Dimethylmercury

This stuff kills slowly and inevitably. A single drop through latex gloves ended a brilliant career in 1997.
The victim didn’t even notice the exposure until months later, when neurological symptoms appeared. By then, it was already too late.
Azidoazide Azide

You might think chemistry follows predictable rules until you encounter a compound that explodes when you look at it wrong (and chemists have tried looking at it from every conceivable angle, with predictably explosive results). This nitrogen-heavy molecule doesn’t just dislike disturbance—it actively resents existing in the first place, which becomes apparent the moment anything more stimulating than a gentle thought occurs in its vicinity.
Even the most experienced explosives researchers treat it like a sleeping dragon with a particularly short temper and an unlimited supply of spite.
So naturally, someone had to make more of it. And naturally, it kept exploding.
But that’s exactly what makes it scientifically fascinating—a compound that challenges the very notion of chemical stability, forcing researchers to reconsider what they thought they knew about molecular bonds and the delicate balance that keeps most substances from spontaneously destroying themselves and everything nearby.
Fluoroantimonic Acid
There’s something deeply unsettling about a substance that makes hydrochloric acid look gentle by comparison. This superacid doesn’t just dissolve materials—it tears apart their molecular structure with the methodical efficiency of a master craftsman dismantling a poorly built house.
Glass containers become suggestions rather than barriers, and even the fumes carry enough corrosive power to eat through materials that most acids wouldn’t dare approach.
The relationship between hydrogen fluoride and antimony pentafluoride that creates this compound feels almost vindictive, as if two already dangerous substances decided to combine forces and see exactly how much destruction they could accomplish together. Researchers working with it don’t just wear protective equipment—they essentially build fortresses around themselves, knowing that the smallest exposure could rewrite their understanding of pain in ways that medical textbooks struggle to adequately describe.
Chlorine Trifluoride
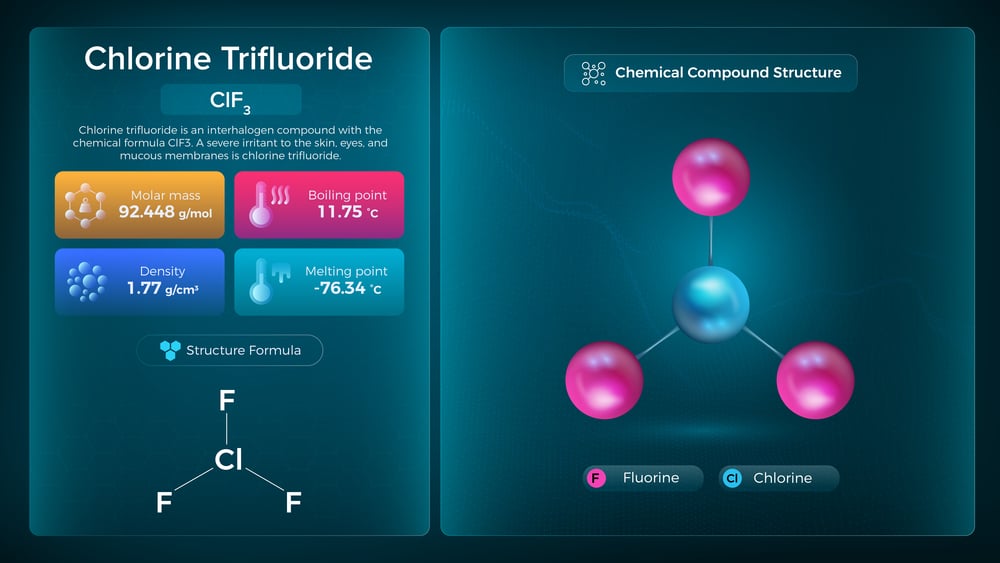
Chlorine trifluoride makes fire seem quaint. It burns through sand, concrete, and asbestos—materials that laugh at ordinary flames.
During World War II, even weapons developers concluded it was too dangerous to weaponize.
When people designing bombs think something is too volatile, that should tell you something about its character.
To be fair, it does have legitimate industrial uses. Which is saying something, considering it ignites everything from water to human tissue on contact.
Batrachotoxin
This alkaloid transforms the simple act of nerve communication into a runaway train with no brakes and no concept of stopping. Found naturally in certain poison dart frogs, the synthetic versions created in modern labs represent one of chemistry’s most elegant approaches to biological chaos—a compound that understands the human nervous system well enough to completely hijack its most fundamental processes.
The indigenous peoples of Colombia figured out its potential centuries ago, but modern researchers have refined it into something far more potent and terrifying. It doesn’t simply block nerve signals or overstimulate them—it forces sodium channels to remain permanently open, turning the body’s own electrical system against itself in a cascade of uncontrollable neural firing.
What makes this particularly unsettling is its precision. This isn’t crude destruction—it’s sophisticated interference with biological machinery, the molecular equivalent of someone who knows exactly which wire to cut to bring down an entire power grid.
Ricin

Protein synthesis stops dead when ricin enters the picture. This toxin doesn’t negotiate or compromise. It shuts down cellular machinery with mechanical precision.
Two milligrams will kill an adult human—about the size of a few salt crystals.
Extracting it from castor beans is disturbingly straightforward. That accessibility makes it a perpetual concern for security agencies worldwide.
Novichok Agents
The Cold War produced many terrible legacies, but few as coldly efficient as the Novichok series—nerve agents designed not just to kill, but to kill in ways that would serve as unmistakable signatures of their origin (a feature that has proven disturbingly relevant in recent international incidents, where the choice of poison carried as much political weight as the act itself). These compounds represent the pinnacle of weaponized chemistry, engineered to be undetectable by standard methods and lethal in quantities so small they challenge the imagination.
But what’s truly chilling about Novichoks isn’t just their lethality—it’s their persistence and the calculated cruelty built into their molecular structure. They bind irreversibly to acetylcholinesterase, the enzyme responsible for cleaning up neurotransmitter signals, essentially turning the body’s own communication system into a jammed radio that keeps broadcasting distress signals until the biological infrastructure collapses entirely.
And they linger, remaining dangerous long after initial exposure, creating contaminated zones that pose risks to anyone who enters without proper protection.
The fact that these compounds were developed in secret, with antidotes remaining classified or nonexistent, reveals something deeply disturbing about the intersection of scientific capability and political intention—chemistry in service of not just killing, but sending messages that transcend the immediate victim.
Dioxin
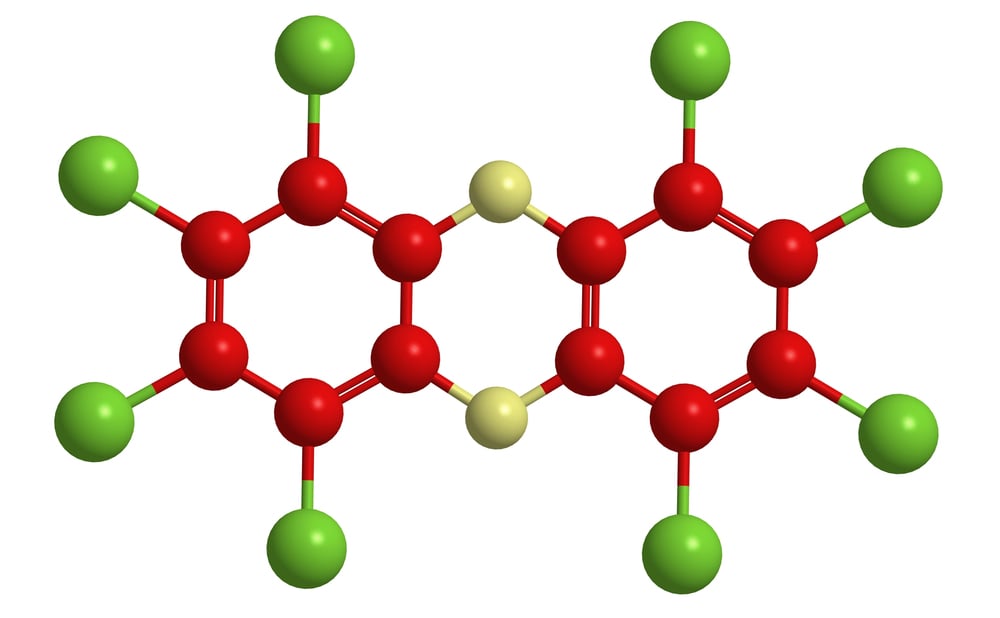
Stability becomes a curse when the compound refuses to break down for decades. Dioxin doesn’t just contaminate—it persists, bioaccumulates, and continues poisoning long after the initial exposure.
It’s chlorine chemistry at its most stubborn and destructive.
The molecular structure looks almost innocent on paper. The reality is anything but innocent.
Botulinum Toxin
One gram could theoretically kill a million people, which places botulinum toxin in the running for the most lethal substance known to science—a distinction that becomes even more unsettling when you consider that we routinely inject diluted versions into foreheads to smooth wrinkles, creating one of modern medicine’s more darkly ironic juxtapositions. This protein, produced by Clostridium botulinum bacteria, doesn’t simply interfere with nerve function—it performs surgical strikes on the molecular machinery that allows neurons to communicate with muscles.
The precision is almost artful. Botulinum toxin cleaves specific proteins involved in neurotransmitter release, essentially cutting the communication lines between brain and body with the accuracy of a master surgeon who knows exactly which connections to sever.
Paralysis follows with methodical inevitability, starting with the muscles controlled by the cranial nerves and progressing downward until respiratory failure becomes inevitable.
What makes this particularly fascinating from a purely scientific perspective is its selectivity—botulinum toxin targets cholinergic synapses specifically, leaving other neural pathways largely intact. The victim remains conscious and aware while their body gradually stops responding to their brain’s increasingly frantic commands.
VX Nerve Agent
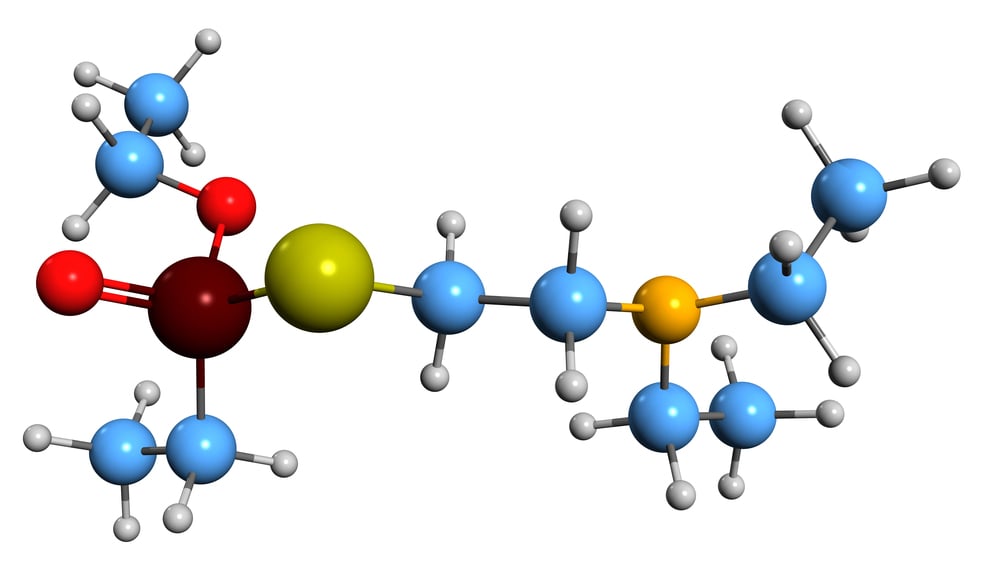
VX represents chemical warfare perfected. This organophosphate compound is designed for maximum lethality with minimum detectability.
A few milligrams on the skin will shut down the nervous system within minutes. It’s colorless, odorless, and persistent.
The molecular structure is deceptively simple. The biological effects are anything but simple.
Strychnine
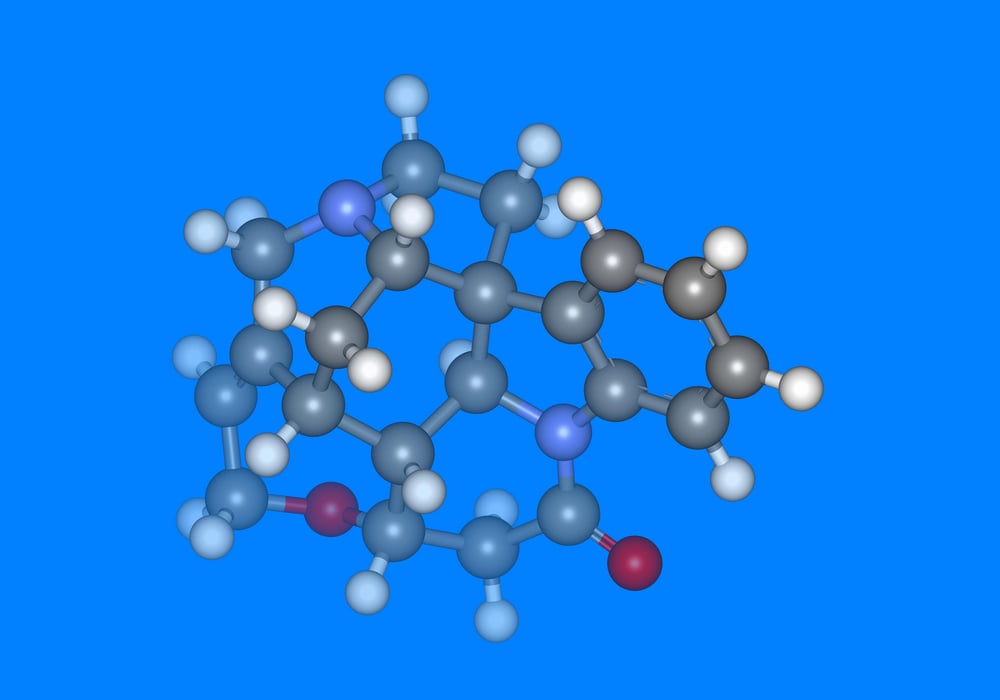
Inhibitory signals in the nervous system exist for good reason—they prevent muscles from contracting simultaneously and creating the kind of full-body seizures that can literally break bones from the inside out. Strychnine blocks these inhibitory signals with ruthless efficiency, turning the body’s own strength against itself in a series of increasingly violent convulsions that represent one of the most physically traumatic ways to die that chemistry has managed to devise.
The compound binds to glycine receptors in the spinal cord, effectively cutting the brake lines on muscular response. What follows isn’t just uncontrolled movement—it’s the body attempting to perform every possible muscle contraction at once, creating a state of tetanic seizure that can be triggered by the slightest stimulus: a sound, a touch, even changes in light.
Amatoxin
Mushroom toxins teach a particularly cruel lesson about delayed consequences. Amatoxins allow their victims to feel perfectly fine for hours, sometimes even days, while quietly dismantling liver and kidney function at the cellular level.
By the time symptoms appear, the damage is typically irreversible.
These cyclic peptides resist cooking, drying, and most other attempts at neutralization. They’re as persistent as they are patient.
Tetrodotoxin
The pufferfish has spent millions of years perfecting a compound that blocks sodium channels with surgical precision. Tetrodotoxin doesn’t destroy nerves—it simply prevents them from firing, creating a state of conscious paralysis where the mind remains trapped in an unresponsive body.
Death comes from respiratory failure while the victim remains fully aware of what’s happening. It’s molecular cruelty refined by evolution into something approaching art.
Polonium-210

Radioactive elements occupy a special category of toxicity because they combine chemical poisoning with radiation damage in ways that overwhelm the body’s repair mechanisms. Polonium-210 is particularly insidious—it emits alpha particles that don’t penetrate skin but wreak havoc once internalized, essentially irradiating organs from the inside out while producing chemical toxicity that compounds the damage.
The half-life of 138 days means victims face months of progressive deterioration as their cellular machinery breaks down under the constant bombardment of radioactive decay. Hair falls out, immune systems collapse, and organs begin failing in sequence as the polonium continues its methodical destruction of biological processes.
What makes this especially terrifying is its invisibility—no immediate symptoms, no obvious warning signs, just the quiet certainty of cellular damage accumulating beyond the point of repair. By the time the effects become apparent, the outcome is already determined.
The Weight of Such Knowledge

Understanding these compounds forces a reckoning with the double-edged nature of scientific progress. Each represents years of careful research, sophisticated equipment, and brilliant minds working to expand human knowledge—yet the result is an arsenal of substances so dangerous that their very existence raises profound questions about the responsibilities that come with such capabilities.
The same labs that create these compounds also work on their antidotes, the same researchers who synthesize them also develop protective measures, and the same knowledge that makes them possible also makes defense against them conceivable.
More from Go2Tutors!

- The Romanov Crown Jewels and Their Tragic Fate
- 13 Historical Mysteries That Science Still Can’t Solve
- Famous Hoaxes That Fooled the World for Years
- 15 Child Stars with Tragic Adult Lives
- 16 Famous Jewelry Pieces in History
Like Go2Tutors’s content? Follow us on MSN.



