Substances With Unusual Chemical Properties
Out there, certain materials act like they’re ignoring what we thought were solid laws of nature. It isn’t magic – just hard proof piling up in labs, leaving scientists scratching their heads even now.
It gets weirder the longer you stare. Fifteen materials sit here breaking every rule folks assume is solid.
Water’s Density Flip

Water behaves oddly compared to most fluids when chilled. While cooling usually packs molecules tighter, water stops doing that past a certain point.
Around 39 degrees Fahrenheit, it becomes as compact as it can get. After that, further cooling makes it swell instead of shrink.
This shift means solid ice ends up lighter than liquid water. So it rests on surfaces instead of sinking below.
Lakes begin freezing at their surface layers first. The deeper parts stay insulated by what’s already frozen above.
Creatures beneath still find room to move and survive. If things worked like ordinary substances, winter might seal off entire bodies of water completely.
Gallium’s Melt in Your Hand

Starting at 85.6 degrees Fahrenheit, gallium begins to melt, a number only slightly higher than normal room warmth. Sitting in your hand, it quickly loses shape, flowing like liquid in moments.
This isn’t illusion – gallium is actual metal, found working inside electronics and bright lights. While heating up, it stays stable far longer than most elements manage, stretching from solid to gas across an unusually broad span.
Its range between becoming fluid and turning vapor ranks among the largest known.
Non-Newtonian Cornstarch

Start mixing cornstarch with water using about twice as much starch, then notice how it pours like a liquid if handled gently. Tap it hard though, and suddenly it locks up solid.
Move fast on top of a big batch, say a kiddie pool full, your feet won’t go under. Halt entirely? Down you go.
This shift – thicker under pressure – is named shear thickening by researchers who now test similar fluids in bendable protective gear.
Aerogel’s Near-Air Density

A tiny bit of substance holds up huge loads, even though it seems barely there. Frozen smoke – that’s what some call it when they see the delicate glow under lab lights.
Weight? Almost zero, yet strength hides inside its ghostlike frame. Heat struggles to move through it, blocked more effectively than in nearly anything else made.
Space missions carried it into alien skies, tucked within rover shells and astronaut gear.
Metallic Hydrogen

Pressure squeezes hydrogen – normally a gas – into something metallic. A team at Harvard said they made it for just an instant back in 2017, yet vanished before anyone learned much.
Should it stay solid without freezing cold, electricity might flow through it endlessly, meeting no resistance. Power systems across distances? Revolutionized, maybe.
But only if someone keeps it stable long enough.
Helium’s Refusal to Freeze

Most things turn solid when cold, yet helium refuses. Near absolute zero, under everyday air pressure, it remains fluid.
To force it into ice form requires intense squeezing during chilling. That happens due to restless movement inside its atoms – zero-point energy – which prevents settling into fixed positions.
Only crushing weight can overcome that jitter.
Dilatant Quicksand

Truth is, real quicksand won’t drown you down like in films. When pushed fast, it fights back – thicker, denser, alive almost.
Panic makes it worse; thrashing only locks you deeper. Calm motion works best, though that feels wrong when fear hits.
Most expect a tug beneath, but stillness wins where force fails.
Fluoroantimonic Acid
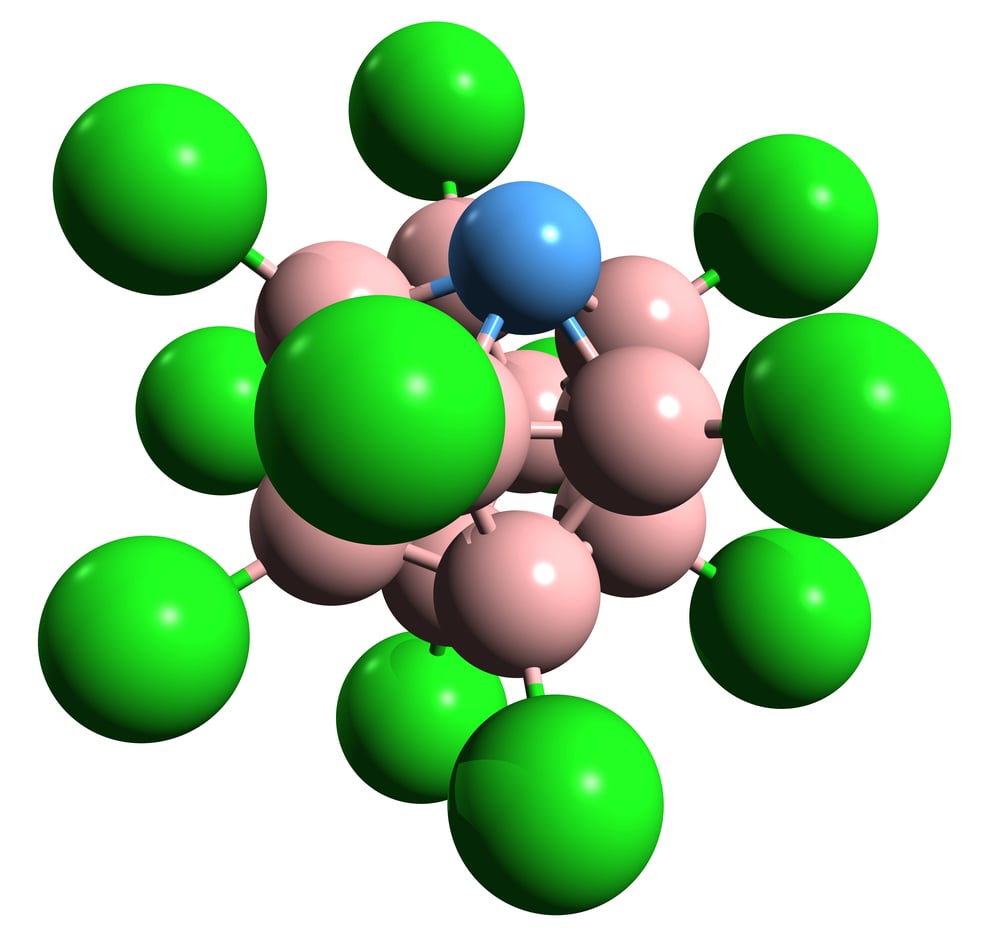
This is the strongest known acid in the world, roughly 20 quintillion times stronger than pure sulfuric acid. It reacts violently with water and eats through glass instantly.
The only containers that can hold it are made of Teflon, because fluorine-based compounds are some of the few things it cannot destroy. Even a small exposure to skin would cause severe damage almost immediately.
Osmium’s Extreme Density

Osmium is the densest naturally occurring element on Earth. A piece the size of a baseball would weigh about 13 pounds.
Its density is so extreme that it competes with iridium for the title of heaviest natural element, depending on how precisely you measure. Despite this, it is brittle enough to be powdered by hand.
Ferrofluid’s Magnetic Response

Ferrofluid is a liquid that becomes strongly magnetized when placed near a magnet. It was originally developed by NASA as a way to move fuel in zero gravity.
The fluid forms dramatic spike-like shapes along magnetic field lines, a phenomenon called the Rosensweig instability. Today it is used in loudspeakers, computer hard drives, and medical research.
Oobleck-Like Starch Under Sound
When oobleck (the same cornstarch mix) is placed on a speaker and exposed to sound waves, it forms moving, finger-like structures that seem to dance. These are called ‘standing waves’ interacting with the non-Newtonian properties of the substance.
The result looks almost alive. Researchers have used this effect to study how pressure waves interact with complex fluids.
Transparent Aluminum

Aluminum oxynitride, sometimes called ‘transparent aluminum,’ is a ceramic compound that is as clear as glass but nearly as hard as ruby. It can stop a .50-caliber bullet.
It is also far lighter than traditional bulletproof glass of the same thickness. The U.S. military has been researching it for use in armored vehicle windows and aircraft.
Bismuth’s Staircase Crystals

When bismuth solidifies slowly from a molten state, it forms hollow, staircase-like crystals with iridescent rainbow colors on the surface. The colors are not dyes or coatings but the result of a thin oxide layer that refracts light.
Each crystal is geometrically precise in a way that looks more designed than natural. Bismuth is also one of the few metals that expands slightly when it freezes, like water.
Supercooled Water

Pure water, free from dust and impurities, can remain liquid well below 32 degrees Fahrenheit without freezing. This is called supercooling.
The moment it is disturbed, touched, or exposed to a tiny particle, it freezes almost instantly in a dramatic chain reaction. Scientists have supercooled water to as low as minus 40 degrees Fahrenheit in carefully controlled lab conditions.
Sulfur Hexafluoride’s Voice Effect

Sulfur hexafluoride is a colorless, odorless gas that is about five times heavier than air. Inhaling a small amount makes the voice go very deep, the exact opposite of helium’s high-pitched effect.
This happens because sound travels more slowly through the dense gas. It is also used as an electrical insulator in high-voltage equipment, and it is actually one of the most potent greenhouse gases known.
Chemistry Is Still Full of Surprises

Science has mapped out most of the periodic table and documented millions of compounds, yet substances like these keep reminding researchers that nature still has curveballs to throw. Gallium melting in a hand, water defying its own freezing rules, and aerogel holding up a brick while weighing almost nothing are not anomalies to be dismissed but clues pointing to deeper principles not yet fully understood.
For anyone who ever found chemistry class a little dry, these substances are proof that the subject has always been far stranger and more entertaining than any textbook suggests.
More from Go2Tutors!

- The Romanov Crown Jewels and Their Tragic Fate
- 13 Historical Mysteries That Science Still Can’t Solve
- Famous Hoaxes That Fooled the World for Years
- 15 Child Stars with Tragic Adult Lives
- 16 Famous Jewelry Pieces in History
Like Go2Tutors’s content? Follow us on MSN.



