17 Medical Devices That Were Banned After Injuries
Medical devices are supposed to heal, not harm. We trust that the gadgets and instruments used in hospitals and clinics have been thoroughly tested and approved for safe use. Unfortunately, the history of medicine is littered with devices that seemed promising but turned out to cause more problems than they solved.
When these devices start injuring patients instead of helping them, regulatory agencies step in to pull them from the market. Here is a list of 17 medical devices that were banned after causing serious injuries to patients.
Dalkon Shield IUD

The Dalkon Shield intrauterine device became one of the most notorious medical device disasters in history. This birth control device was linked to severe pelvic infections, infertility, and even death in some cases. The device’s multifilament tail string acted like a wick, allowing bacteria to travel from the vagina into the uterus, causing life-threatening infections.
Bjork-Shiley Heart Valve
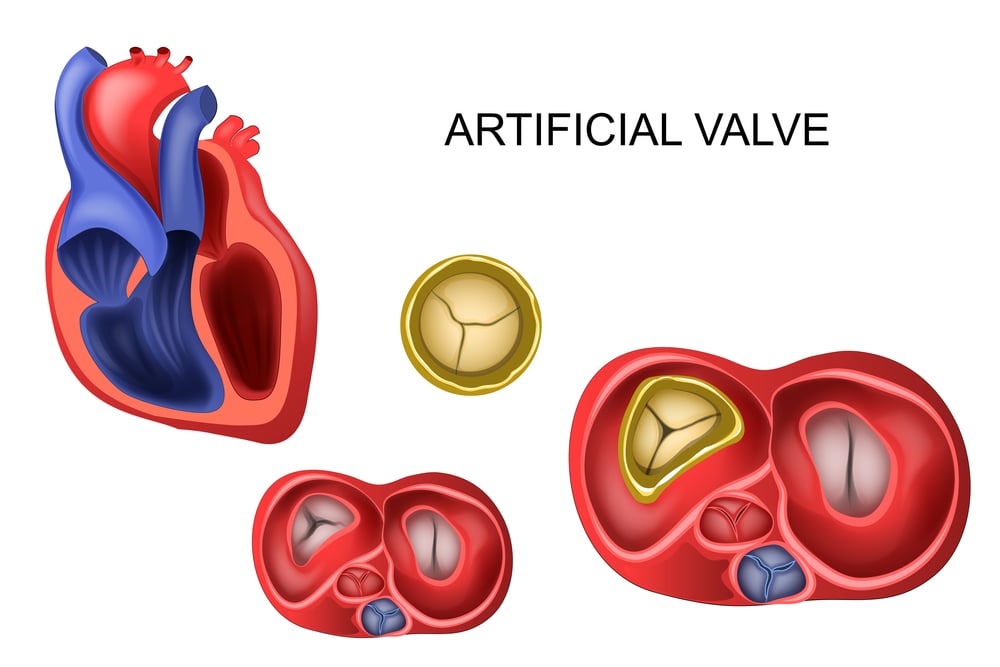
This artificial heart valve seemed like a miracle of modern engineering until it started fracturing inside patients’ chests. When the valve’s metal struts broke, it often resulted in catastrophic heart failure and death. The fractures were unpredictable and could occur years after implantation, making it impossible for doctors to know which patients were at risk.
Sulzer Hip Implants

These hip replacement devices were supposed to last for decades, but instead they began failing within months of implantation. The problem was with the manufacturing process, which left oil residue on the implants that prevented proper bone growth. Patients experienced severe pain, loosening of the implant, and required additional surgeries to remove and replace the faulty devices.
Poly Implant Prothèse Breast Implants

French company PIP used industrial-grade silicone instead of medical-grade material in their breast implants to cut costs. This cheaper silicone was more likely to leak and rupture, causing inflammation, pain, and potential health risks. When the scandal broke, hundreds of thousands of women worldwide had to have their implants removed.
Fentanyl Patches by Duragesic

While fentanyl patches themselves weren’t banned, certain defective batches were recalled after causing fatal overdoses. The patches were leaking excessive amounts of the powerful painkiller through faulty seals, delivering lethal doses to unsuspecting patients. Some people died from fentanyl poisoning even when using the patches exactly as prescribed.
Meridia Weight Loss Drug

Though technically a medication rather than a device, Meridia’s delivery system and monitoring requirements classified it as a medical device program. This appetite suppressant was pulled from the market after studies showed it significantly increased the risk of heart attacks and strokes. The cardiovascular risks outweighed any potential weight loss benefits.
Duofilm Wart Remover Patches
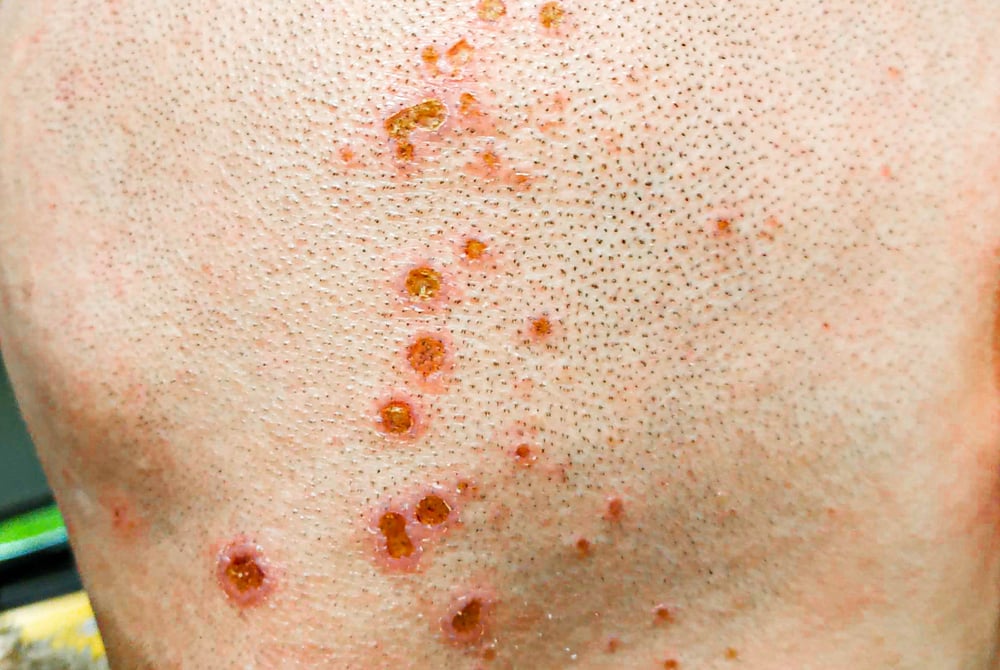
These over-the-counter wart removal patches contained salicylic acid that was too concentrated for safe home use. Many users experienced severe chemical burns, permanent scarring, and tissue damage. The patches were particularly dangerous when used on sensitive skin areas or left on longer than recommended.
Zicam Nasal Spray
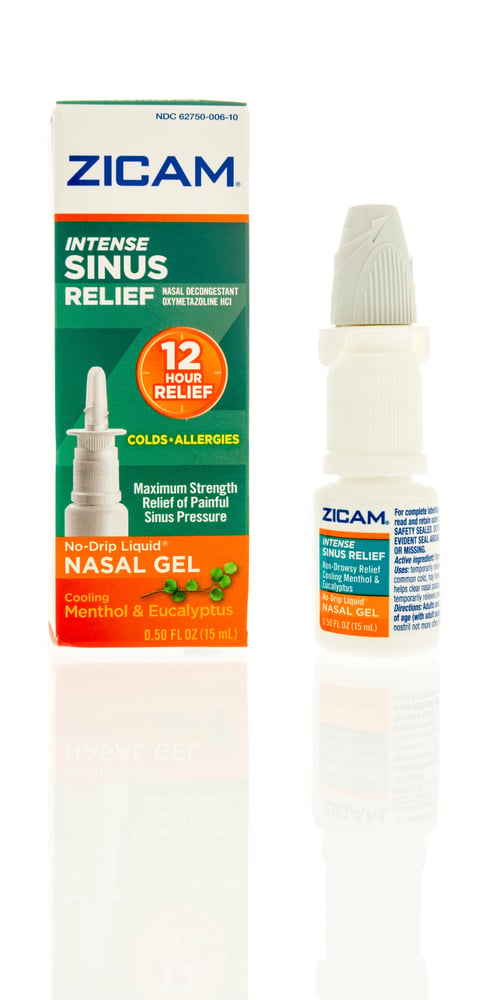
This cold remedy was supposed to shorten the duration of cold symptoms, but it ended up destroying users’ sense of smell permanently. The zinc-based formula damaged the olfactory nerves in the nose, leaving many people unable to taste or smell anything. The loss of these senses was often permanent and irreversible.
Essure Birth Control Implants
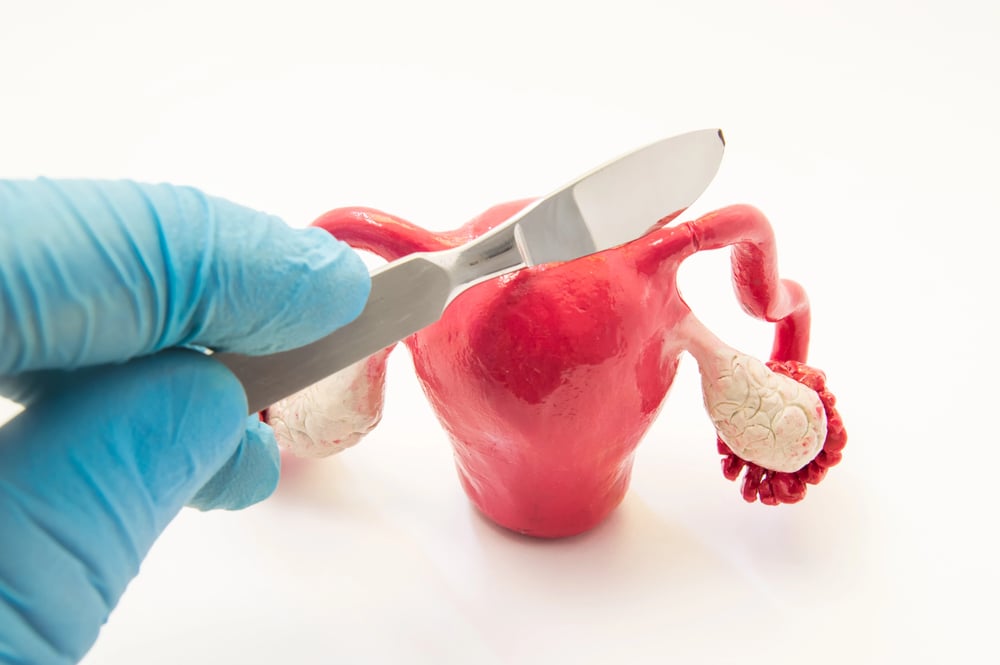
These small metal coils were inserted into women’s fallopian tubes as a permanent form of birth control. However, the devices often migrated from their intended location, perforated organs, and caused chronic pain. Many women required emergency surgery to remove the devices, and some suffered permanent damage to their reproductive systems.
Accutron Pacemaker
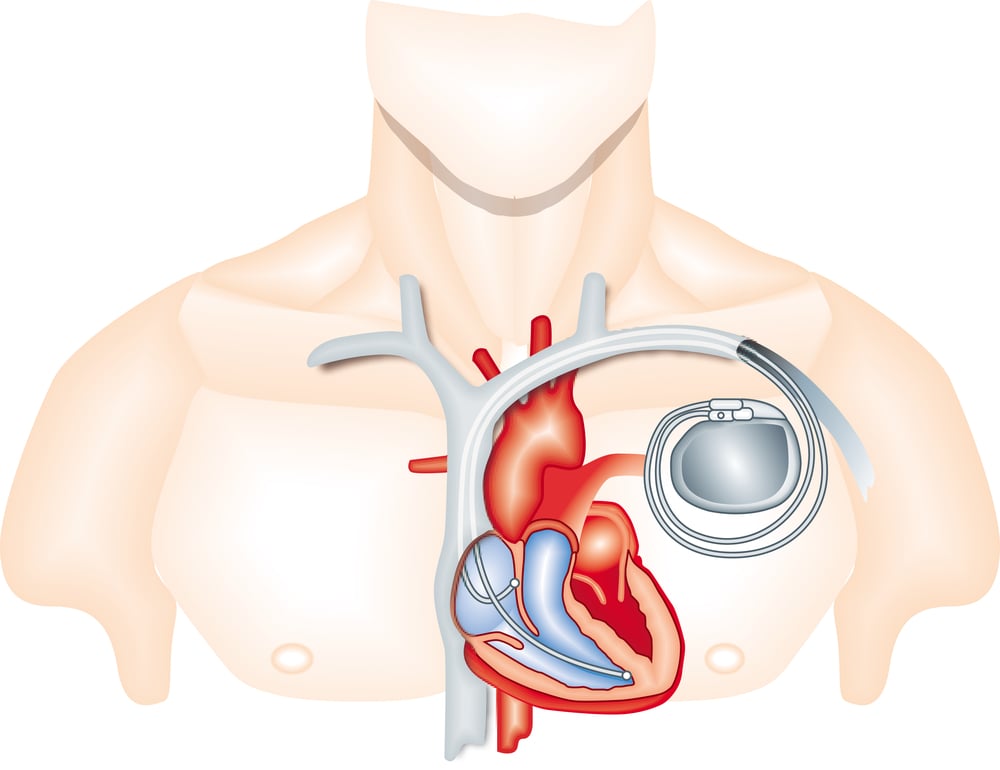
This early pacemaker model had a fatal flaw in its battery design that caused unexpected failures. When the battery died suddenly, patients could experience cardiac arrest without warning. The unpredictable nature of the failures made it impossible for doctors to monitor patients effectively or replace the devices before they failed.
Orthopedic Bone Growth Stimulators
Certain models of these devices, designed to help bones heal faster, were found to emit dangerous levels of electromagnetic radiation. Patients using these stimulators experienced tissue burns, nerve damage, and interference with other medical devices like pacemakers. The radiation exposure was far higher than considered safe for extended use.
Thermacool Radiofrequency Device

This cosmetic device was marketed for skin tightening and wrinkle reduction but caused severe burns and permanent scarring in many patients. The radiofrequency energy was difficult to control, leading to unpredictable heating of skin tissue. Some patients required skin grafts to repair the damage caused by the device.
Relisys Glucose Monitoring System
This blood glucose monitor for diabetics gave inaccurate readings that led to dangerous medication errors. Patients received false low or high readings, causing them to take incorrect doses of insulin. Some diabetics went into diabetic coma or experienced severe hypoglycemia because they trusted the faulty readings.
Hydroxyapatite Breast Implants

These implants were made from a ceramic material that was supposed to be more natural than silicone. However, the ceramic particles would break down and migrate throughout the body, causing inflammation and granulomas. The particles were impossible to remove completely once they spread, leading to chronic health problems.
Magnetic Resonance Therapy Devices
These devices claimed to treat various conditions using magnetic fields, but they interfered dangerously with pacemakers and other implanted medical devices. Several patients with pacemakers experienced device malfunctions and cardiac events when exposed to these magnetic therapy devices. The magnetic fields were strong enough to reprogram or disable life-supporting implants.
Vaginal Mesh Implants

These surgical mesh products were used to treat pelvic organ prolapse and incontinence but caused severe complications in many women. The mesh would erode through vaginal tissue, cause chronic pain, and lead to infections. Many patients required multiple surgeries to remove the mesh, and some suffered permanent damage to their pelvic organs.
Infuse Bone Graft
This bone growth protein was supposed to help spinal fusion surgery heal faster, but it caused dangerous swelling and inflammation. When used in neck surgery, the protein caused throat swelling that could block breathing and require emergency tracheotomy. The inflammation also increased the risk of infection and delayed healing rather than promoting it.
Learning From Medical Mistakes

These banned devices serve as sobering reminders that medical innovation comes with risks that aren’t always apparent during initial testing. Each of these failures led to stricter regulations, better testing protocols, and more rigorous oversight of medical devices. While these incidents caused tremendous suffering for patients, they ultimately made modern medicine safer by highlighting the importance of thorough testing and post-market surveillance. The lessons learned from these failures continue to protect patients today.
More from Go2Tutors!

- 16 Historical Figures Who Were Nothing Like You Think
- 12 Things Sold in the 80s That Are Now Illegal
- 15 VHS Tapes That Could Be Worth Thousands
- 17 Historical “What Ifs” That Would Have Changed Everything
- 18 TV Shows That Vanished Without a Finale
Like Go2Tutors’s content? Follow us on MSN.



