Fascinating Facts About the Rarest Earth Metals
You probably have rare earth metals in your pocket right now. There’s a good chance there’s some in your home, your car, and every piece of tech you own.
These 17 elements sit quietly at the bottom of the periodic table, and most people have never heard of a single one. But pull any of them out of the equation, and the modern world starts to fall apart pretty quickly.
The story of these metals — where they come from, what they do, and why they matter so much — is stranger and more consequential than almost anyone realizes.
They’re Not Actually Rare

The name is misleading, and it has been from the start. Rare earth metals got their label in the late 18th and early 19th centuries when scientists first found them in minerals that were, at the time, only known in a handful of locations.
The word “earth” itself is an old term for oxides — the chemical form these elements take in nature. Neither word in the name accurately describes what these metals are today.
Cerium, the most abundant of the group, is the 25th most common element in the Earth’s crust. That makes it roughly as plentiful as copper.
The real challenge isn’t finding them. They almost never appear in concentrated deposits, and they don’t exist as pure metals in nature.
They’re locked inside minerals, mixed together, and separating them from one another is extraordinarily difficult. That’s what makes them hard to work with — not scarcity.
Some of Them Were Born in Dying Stars

Most rare earth metals are heavier than iron, which means they weren’t formed during the Big Bang or through ordinary stellar processes. They were forged inside supernovas — exploding stars — and through a slower process called the s-process that occurs in a specific type of ageing star.
The raw material for these elements was scattered across space billions of years ago and eventually collected into the rocky body that became Earth. This cosmic origin gives rare earths a connection to the deep history of the universe that few other elements on the periodic table can claim.
Separating Them Is One of Chemistry’s Hardest Problems

All 17 rare earth elements share nearly identical chemical properties. They look alike, behave alike in most reactions, and dissolve in similar ways.
For over a century after their initial discovery, chemists couldn’t reliably tell them apart. Early researchers kept thinking they had found a single element, only to discover later that what they had was actually a mix of several.
It took decades of painstaking work — and the eventual development of ion-exchange chromatography — to separate them into individual elements. Even today, the process is technically complex and expensive. For many industrial applications, manufacturers simply use a mixed blend of rare earths rather than bothering to isolate specific ones.
Promethium: The Element That Barely Exists

Of all the rare earths, promethium is the strangest. It has no stable isotopes whatsoever — every single form of it is radioactive. At any given moment, there is estimated to be less than one kilogram of promethium in the entire Earth’s crust. It decays so quickly that any promethium the planet once had has long since vanished.
Promethium was discovered in 1945 at Oak Ridge National Laboratory, making it the last of the rare earth lanthanides to be found. Scientists named it after Prometheus, the Greek Titan who stole fire from the gods — a deliberate nod to the nuclear age and the dangers of human ambition. It wasn’t until 2024 that researchers fully characterized its chemical properties in solution for the first time, filling what had been described as a glaring gap in our understanding of the periodic table.
Iridium Helped Solve the Mystery of the Dinosaurs

Iridium is one of the rarest metals on Earth, occurring at roughly 0.3 parts per billion in the crust. But it’s far more abundant in asteroids. That difference turned out to be one of the most important clues in the history of science.
In the late 1970s, geologist Walter Alvarez and his father, Nobel Prize-winning physicist Luis Alvarez, discovered an abnormally high concentration of iridium in a thin layer of clay found worldwide at the geological boundary marking the end of the age of dinosaurs, 66 million years ago. The iridium levels at that boundary were more than 20 times higher than normal in some locations. Because iridium is so rare on Earth but so common in space rocks, the Alvarezes proposed that a massive asteroid had struck the planet, vaporized, and scattered iridium across the globe. The theory was controversial at first, but it eventually became the most widely accepted explanation for the mass extinction that wiped out 75% of life on Earth. The impact crater, known as Chicxulub, was later found off the coast of Mexico’s Yucatán Peninsula.
Some of Them Burst Into Flames on Their Own

A handful of rare earth metals are pyrophoric in finely divided form — meaning they can ignite spontaneously when exposed to air. Terbium and ytterbium are among those that behave this way. All rare earths also tarnish quickly when exposed to air at room temperature and react with water to produce hydrogen gas.
This reactivity is part of why these metals are so difficult to handle in their pure form and why they’re almost always processed and used as compounds rather than raw metals.
The Strongest Magnets on Earth Depend on Them

Neodymium magnets — made from neodymium, iron, and boron — are the most powerful permanent magnets ever created. They’re the reason your smartphone is as small as it is, the reason electric vehicles can run efficiently, and the reason wind turbines can generate the power they do. Without rare earth magnets, none of these technologies would work in their current form.
In 2021, permanent magnets accounted for the largest share of global rare earth consumption — about 29% of all use. The demand is only growing as the world pushes toward cleaner energy and smaller, more powerful devices.
They Fight Counterfeiting
One of the more unexpected uses of rare earth metals is in anti-counterfeiting technology. Dysprosium, europium, and terbium are all fluorescent under ultraviolet light. When tiny amounts of these elements are blended into genuine products — banknotes, documents, luxury goods — they become invisible markers. Hold a UV light over the product and the rare earths glow. Hold it over a fake and nothing happens.
It’s a surprisingly elegant application for elements that most people associate with heavy industry.
Extracting Them Creates a Staggering Amount of Waste

Mining rare earths is an environmental nightmare. Estimates suggest that extracting one metric ton of rare earth elements produces roughly 2,000 metric tons of waste, some of it toxic and about one ton of it radioactive. The largest rare earth mining operation in the world, Bayan Obo in China, has generated over 70,000 tons of radioactive waste that has contaminated local groundwater.
The environmental cost of these metals is one of the reasons why Western countries historically pushed this kind of mining elsewhere rather than dealing with it on their own soil. That decision is now coming back to haunt them.
China Controls Almost Everything
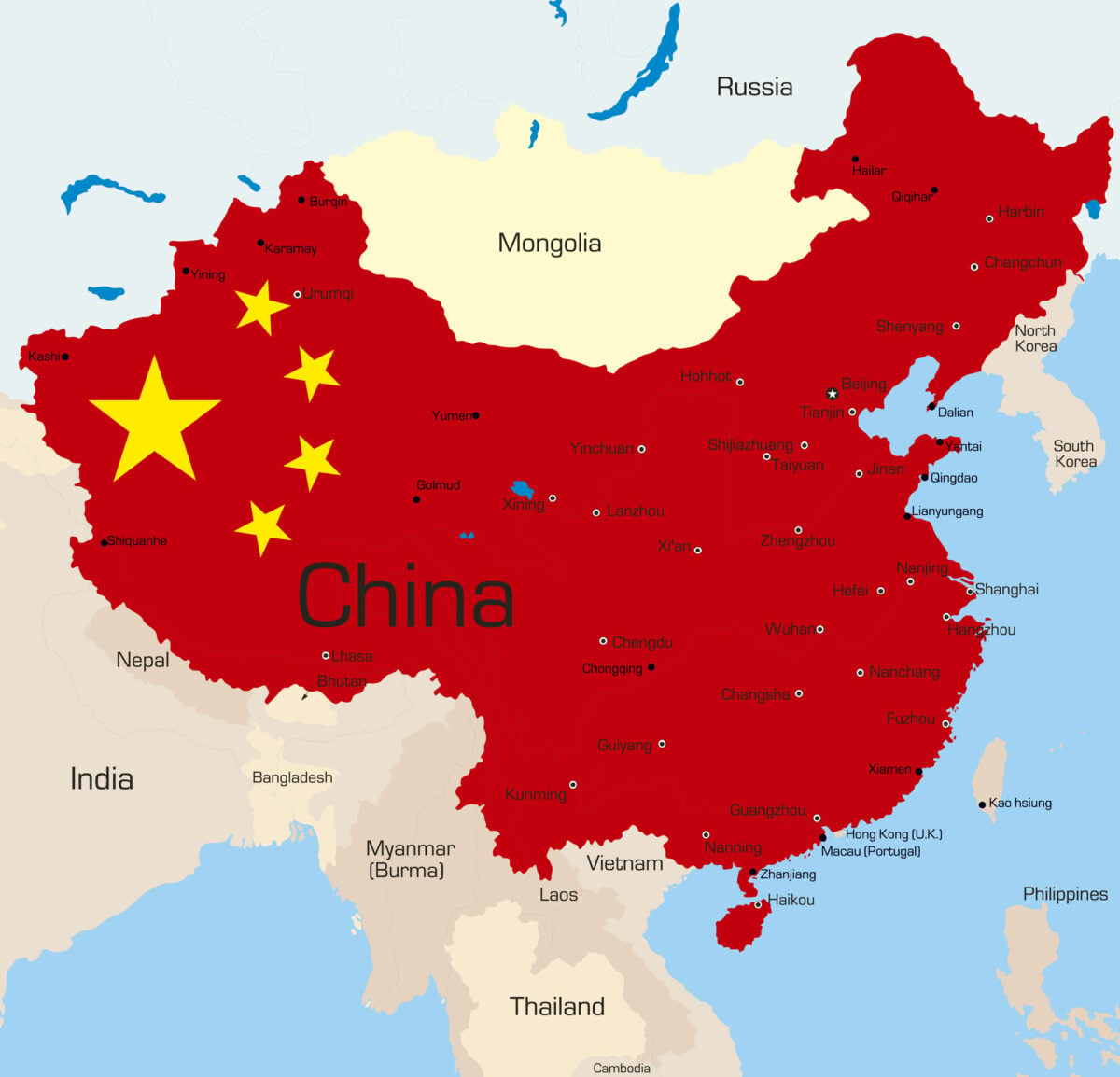
China dominates the rare earth supply chain to a degree that is difficult to overstate. As of 2024, China accounted for roughly 60% of global rare earth mining and about 91% of the world’s separation and refining capacity. For rare earth permanent magnets — the highest-value product in the chain — China controls approximately 93% of global manufacturing.
This dominance was not accidental. Beginning in the 1990s, China pursued a deliberate industrial policy to build out its rare earth capabilities from mining all the way through to finished magnets. China’s then-leader Deng Xiaoping reportedly summed up the strategy simply: “The Middle East has oil; China has rare earth elements.”
They’re at the Center of a Geopolitical Standoff

In 2025, rare earths became one of the flashpoints of the trade war between the United States and China. After the U.S. escalated tariffs on Chinese goods, China responded by introducing export restrictions on seven rare earth elements. U.S. manufacturers scrambled to secure supplies, and Ford temporarily shut down its Chicago factory due to shortages of rare earth magnets.
Rare earths are critical to defense technologies as well — they appear in F-35 fighter jets, submarine guidance systems, Tomahawk missiles, and radar equipment. The U.S. currently has only one manufacturer of rare earth magnets on domestic soil. The vulnerability is real, and governments on both sides of the Pacific are acutely aware of it.
Recycling Could Change Everything — But It Hasn’t Yet

The rare earths in consumer products can, in theory, be recovered and reused. A single neodymium magnet from a hard drive or an electric motor contains enough material to be worth recycling. Nickel-metal hydride batteries hold grams to kilograms of rare earth alloys. These materials contain 25 to 30 percent rare earths by weight — far more concentrated than even the best ore deposits.
But in practice, recycling rates for rare earths remain extremely low. Consumer electronics contain only tiny amounts of the metals — a fraction of a gram in a phone’s speaker magnet, for instance — and the complexity of modern devices makes separating them out difficult. Until the economics and the technology catch up, most rare earths end up in landfills rather than back in the supply chain.
They May Become Even More Critical Than They Are Now

A study from the Belgian research university KU Leuven concluded that Europe alone would need seven to 26 times more rare earth metals than it currently uses if it wants to hit its climate targets by 2050. The International Energy Agency offered a more conservative estimate, predicting that global demand could be three to seven times higher by 2040 than it is today.
The energy transition — the shift from fossil fuels to renewables — depends on rare earths in ways that are often invisible to the public. Wind turbines, electric vehicle motors, solar panels, and energy-efficient lighting all require them. The metals are classified as critical raw materials by the U.S., the European Union, and the United Kingdom, and for good reason.
Hidden in Plain Sight, Holding Everything Together

Out there on display counters, rare earth metals are nowhere to be seen. Shiny they’re not. Walk into any shop and those plain silvery-grey bars would blend into the background without notice.
Yet quietly present behind almost every piece of today’s world – tucked inside your mobile device, hidden under the hood of vehicles, working through invisible wires that feed electricity where it’s needed. Out here among the remains of exploded suns, these bits first took form.
Trapped now beneath rock and time, they wait. Whoever holds them shifts how nations deal with one another.
Though nearly invisible to daily life, their pull runs deep. Hidden names on the periodic table shape much more than anyone tends to notice.
More from Go2Tutors!

- The Romanov Crown Jewels and Their Tragic Fate
- 13 Historical Mysteries That Science Still Can’t Solve
- Famous Hoaxes That Fooled the World for Years
- 15 Child Stars with Tragic Adult Lives
- 16 Famous Jewelry Pieces in History
Like Go2Tutors’s content? Follow us on MSN.



